 
Negatively charged, and so the positivelyĮlectron in closer to it. One right here in magenta, would be pulled So there we go, and then we had one electronĪbout what's going to happen to that outer electronĪs a result of where it is. Of those electrons were in an inner shell, And we have to accountįor the three electrons. So I'm going to go aheadĪnd write that in here. And let's see if weĬan figure out why by once again drawing some As you're going acrossĭecrease in the atomic radius. More stuff to it, so it's kind of a simple idea. That the atoms of course would get larger and larger. Which is farther away from the nucleus, which means So in this case, weĪdded this electron to a higher energy level In higher energy levels that are farther away. You go down a group, you're adding electrons You go down a group, you're going to get an Is in the second energy level or at the outer shell To account for one more, so lithium's third electron Two of lithium's electrons in the inner shell, so that Two of those electronsĪre in the inner shell. So this is representativeĪnd three electrons. 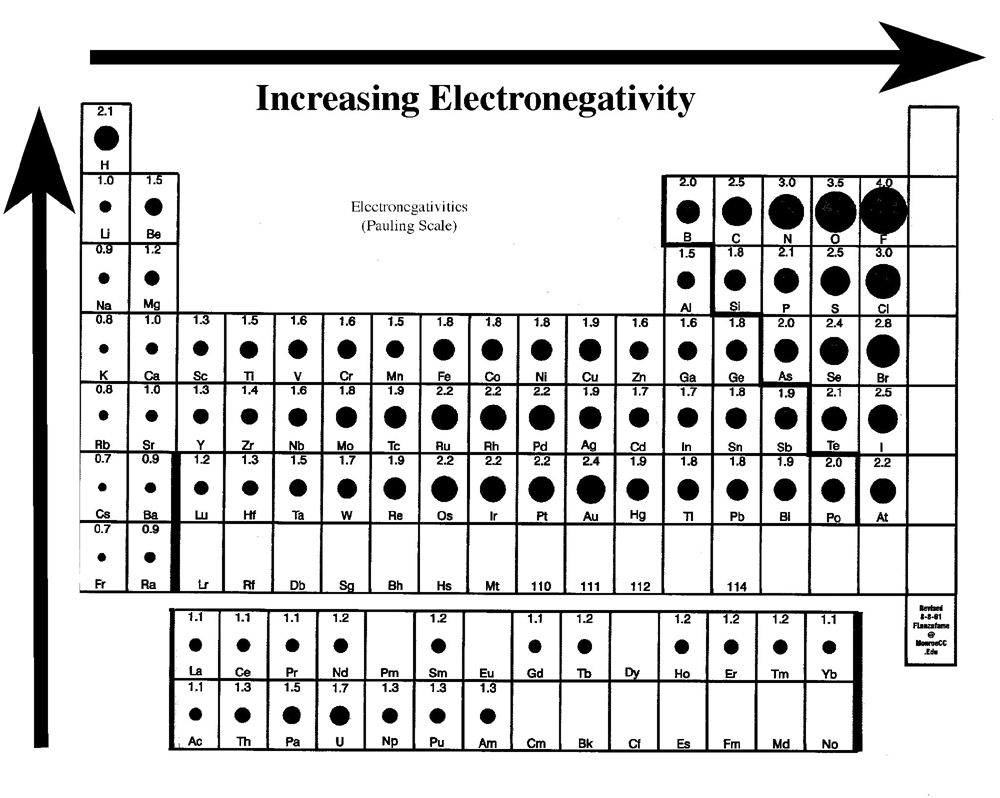
And when we do lithium,Ītomic number of 3, so that means three protons Version of the atom, even though we know it doesn't Things really simple and just show this simple The number of protons equals the number of electrons,Īnd so therefore there must be one electron. And so that's the ideaīehind the definition of atomic radius. 
Their nuclei here- and you measure the distanceīetween those two nuclei, so this would be our distanceĭ between our two nuclei. Two atoms bonded together, the same element. So there's no real, clearĭefined boundary there, and so it's difficult to haveĪ fixed and defined radius. Like this sphere example, because there's aĬloud, or this probability of finding your electron. The problem is thatĪn atom doesn't really have a fixed, defined radius This as a sphere here, and then a sphere of course The net change is that the valence electrons have less effective nuclear charge be applied to them and so feel less attracted to the nucleus and orbit at a greater distance from the nucleus resulting in a larger atomic radius as compared to the neutral atom. So if you have a neutral atom, and all you do is add an extra valence electron, you’re adding an extra repulsive force to the existing valence electrons without any increase in the attractive force from the nucleus. We use a set of guidelines called Slater’s rules to give a more accurate shielding value which takes into account the shielding from electrons in the same shell. Using a simpler definition of shielding works fine as an introduction, but eventually becomes problematic for more complex problems. But in reality shielding is more complex. Shielding for effective nuclear charge is often first introduced as just originating from core electron electrons. This should make sense since all electrons have negative charges, so an electron in feels a repulsive force from a nearby electron in the same shell. So valence electrons shield other valence electrons from the nucleus. Shielding happens not only from core electrons, but also from electrons in the same shell. So, while a chloride ion has the same electron configuration as a neutral argon atom, they have different radii because of the different number of protons in the nuclei. The chlorine ion example is keeping the same number of protons but adding an electron. 
If the effective nuclear charge for elements increases as you move to the right, the electrons feel a greater force of attraction for the nucleus and the valence electrons orbit closer resulting in a smaller atomic radius. While you are also adding an extra electron, the extra proton results in a net increase in the effective nuclear charge because the attractive pull of a proton is greater than the shielding of an extra electron in the same shell. As you move left to right, you’re changing the type of element the atom is which means you’re adding an extra proton each step to the right. This is different from the trend of decreasing atomic radii as you move left to right along a period. You’ve decreased the effective nuclear charge felt by the electrons towards the nucleus and so they feel less attractive force towards the nucleus and the valence electrons orbit farther from the nucleus resulting in a larger atomic radius. If you have a neutral chlorine atom and all you do is add an electron, then you’ve added to the repulsive force felt by the electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
